α2 –plasmin inhibitor (α2 -PI) is a single-chain glycoprotein and is one of the major serine proteinase inhibitors circulating in plasma. Physiologically, it is the predominant inhibitor of plasmin and it therefore plays a significant role in the specific inhibition of fibrinolysis. The role of α2 -PI in fibrinolysis is three fold: covalent inhibition of plasmin; interference with the binding of plasminogen to fibrin; and factor XIIIa catalyzed cross-linking of α2-PI to fibrin (1). Rapid inactivation of plasmin proteolytic activity occurs through a two-step process. The inhibitor first forms a reversible complex with plasmin which is sub-sequently followed by the formation of a covalent, enzymatically inactive, complex with the catalytic site in plasmin (2,3). α2-PI also functions by interfering with the binding of plasminogen to fibrin, effectively slowing the activation of plasminogen by fibrin-bound plasminogen activator (4). The interference in binding ultimately delays the initiation of fibrinolysis. Covalent cross-linking of a2-PI to the a-chains of fibrin which is mediated by factor XIIIa, protects crosslinked fibrin clots from plasmin degradation and thereby markedly stabilizes the fibrin clot against fibrinolysis (5). Failure to protect the fibrin clot from rapid dissolution before injured vessels can be restored results in a bleeding tendency described in patients with a deficiency in α2-PI or factor XIII (6,7).
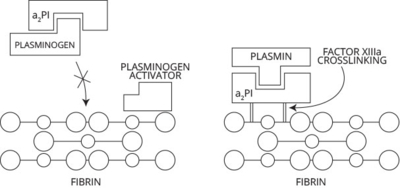
a2–plasmin inhibitor and fibrinolysis
The primary physiological inhibitor of plasmin is a2–plasmin inhibitor. The binding of a2 –plasmin inhibitor to plasmin(ogen) plays a critical role in fibrinolysis. The binding of circulating free plasmin(ogen) by a2 –plasmin inhibitor prevents both zymogen and enzyme from binding to fibrin and ultimately initiating fibrinolysis. a2–Plasmin inhibitor already crosslinked to fibrin prevents plasmin catalytic action on local sites of fibrin deposition as well as impeding subsequent activation of plasminogen via plasminogen activator.
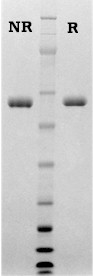
| Gel | Novex 4-12% Bis-Tris |
|---|---|
| Load | Human α2 –Plasmin Inhibitor, 1 µg per lane |
| Buffer | MOPS |
| Standard | SeeBluePlus 2; Myosin (191 kDa), Phosphorylase B (97 kDa), BSA (64 kDa), Glutamic Dehydrogenase (51 kDa), Alcohol Dehydrogenase (39 kDa), Carbonic Anhydrase (28 kDa), Myoglobin Red (19 kDa), Lysozyme (14 kDa) |
| Localization | Plasma | |||||
|---|---|---|---|---|---|---|
| Mode of action | Inhibits plasmin by forming an irreversible complex with the catalytic active site. Prevents the binding of plasmin to fibrin. Cross-linked by factor XIIIa to fibrin. | |||||
| Plasma Concentration | 69 µg/ml (1) | |||||
| Molecular weight | 58,700 single-chain (determined from amino acid sequence and 14% carbohydrate) and 67,000 (determined by SDS-PAGE) (1) | |||||
| Extinction coefficient |
|
|||||
| Isoelectric Point | Unknown | |||||
| Structure | Circulates as single chain molecule consisting of 452 amino acids (1). | |||||
| Percent carbohydrate | 14% (w/w) (1) |
- Aoki, N. et al., Methods Enzymol., 223, 185-197 (1993).
- Moroi, M. and Aoki, N., J. Biol. Chem., 251, 5956 (1976).
- Wiman, B. and Collen, D., J. Biol. Chem., 254, 9291 (1979).
- Moroi, M. and Aoki, N., Thromb. Res., 10, 851 (1977).
- Sakata, Y. and Aoki, N., J. Clin. Invest., 69, 536 (1982).
- Aoki, N. et al., J. Clin. Invest., 63, 877 (1979).
- Israels, E. D. et al., J. Clin. Invest., 52, 2398 (1973).
- Holmes, W. E. et al., J. Biol. Chem., 262, 1659 (1987).
- Kluft, C. and Los, N., Thromb. Res., 21, 65 (1981).
- Tamaki, T. and Aoki, N., J. Biol. Chem., 257, 14767 (1982).
- Tseng, I., et al., Am J Physiol Cell Physiol 295:423-431, 2008
This publication list is not all encompassing, and is only meant to provide limited examples of how Prolytix products are used. We encourage you to search the literature for other examples pertinent to your experimentation, and to contact us with any technical questions.
