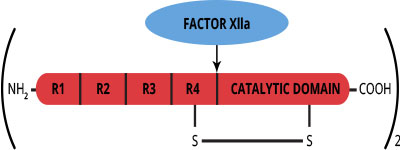
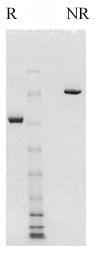
Factor XI is a plasma glycoprotein which circulates in a non-covalent complex with high molecular weight kininogen (1). The mature molecule is synthesized in the liver and is a two-chain homodimer with a molecular weight of approximately 160,000 (2,3). It is estimated that 5% of the total mass is attributable to carbohydrate (2). The two identical monomers have molecular weights of 80,000, and are joined together by disulfide bonds. Thus by SDS-PAGE analysis, factor XI appears as a single band both non-reduced (Mr=160,000), and reduced (Mr=80,000).
Factor XI circulates as a zymogen and requires proteolytic activation to acquire serine protease activity. The conversion of factor XI to factor XIa is catalyzed by factor XIIa, and results in cleavage of the Arg369-Ile370 bond in each monomer (3). Factor XIa consists of two NH2-terminal derived heavy chains, and two COOH-terminal derived light chains, all of which are held together by disulfide bonds. Factor XIa participates within the intrinsic pathway of coagulation by catalyzing the conversion of factor IX to factor IXa. A bleeding disorder called plasma thromboplastin antecedent deficiency results from a lack of factor XI procoagulant activity (4,5). The variable bleeding tendencies observed in factor XI deficient patients do not correlate with either factor XI activity or antigen levels. This latter observation may be related to the ability of the tissue factor/factor VIIa complex to also activate factor IX to IXa.
Historically, factor XI has been difficult to purify due to its relatively low concentration in plasma, and its susceptibility to proteolysis (6). Factor XI is purified from fresh frozen plasma that is stabilized by added inhibitors. The plasma is first treated with BaCl2 to remove the vitamin K-dependent proteins, and factor XI is then isolated by affinity chromatography. A final chromatography step on heparin sepharose yields a homogeneous preparation of intact factor XI. The finished product is supplied in 50% (vol/vol) glycerol/H2O and should be stored at -20oC.