Human blood plasma is rich in proteins, including immunoglobulins (IgG), coagulation factors (Factors II, VIII, IX, and X), alpha-1 antitrypsin, fibrin sealants, and albumin. These proteins boost the immune system and fight inflammation and infection, making plasma-derived biotherapeutics useful in treating rare, chronic, and often genetic diseases. Unfortunately, the current worldwide demand for intravenous IgG (IVIG) products exceeds current plasma collection supply.
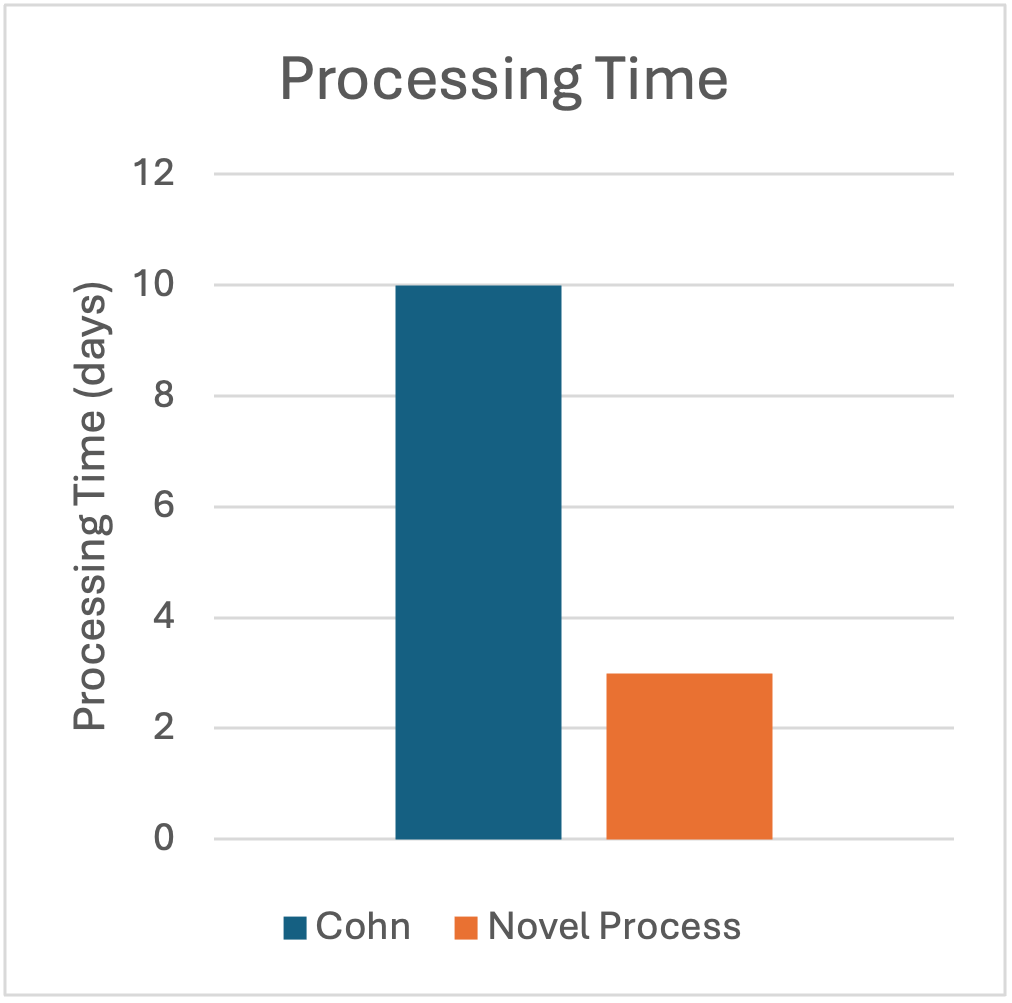
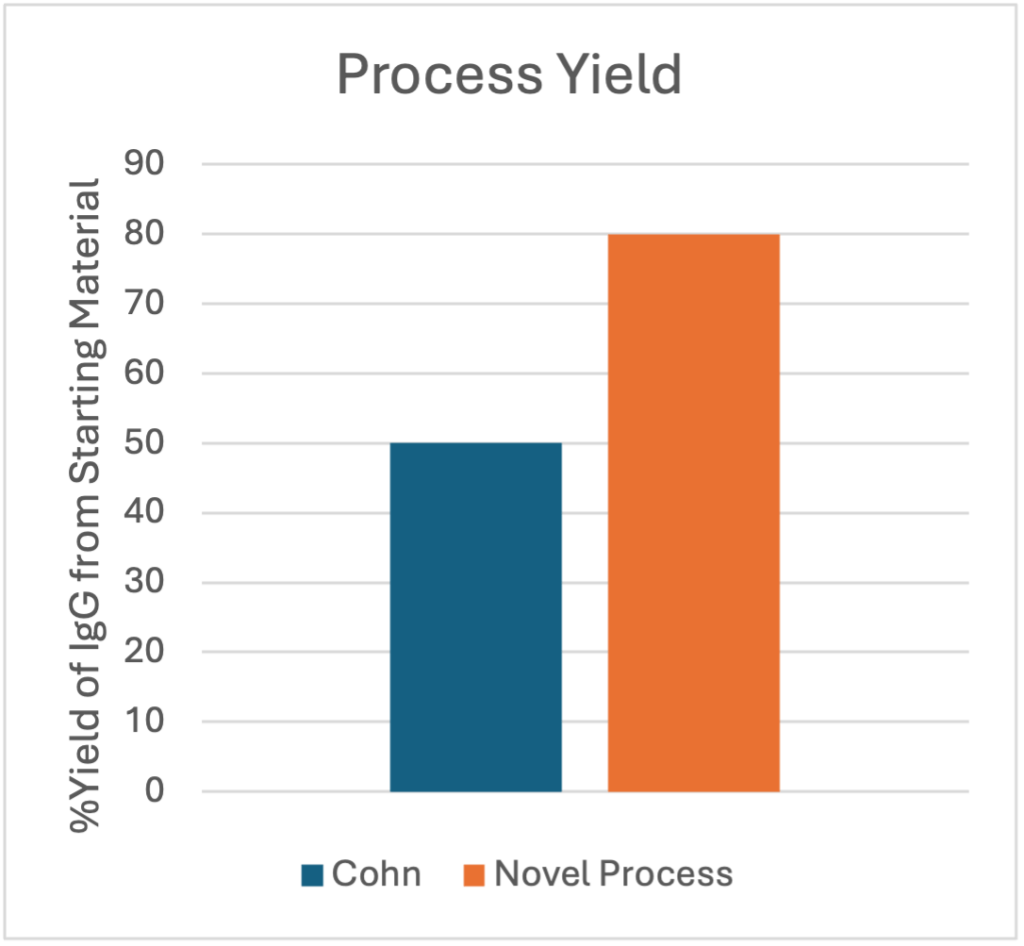
For over 70 years, IgG has been produced using the Cohn process, which fractionates – plasma proteins based on their differential solubility in ethanol with variances in temperature, pH, ionic strength, and protein concentration. The modern Cohn process is widely known to achieve yields of only 50-60% of the starting IgG, and the process takes between 7-10 days to complete. Not only does working with large volumes of ethanol create environmental concerns, IgG produced by Cohn fractionation also carries an inherent risk of protein denaturation, because it is produced with the combination of alcohol and extreme pH changes.
Avoiding the combined effects of alcohol and low pH could improve protein stability, in vivo half-life, patient tolerability, and reduce the immunogenicity of protein therapeutics. Given the scarcity of IgG products and the inability of plasma collections to satisfy patient demands for IgG, there is a clear need for a new process that addresses the shortcomings of the Cohn fractionation process.
Procoagulant Contaminants in Plasma-derived Biotherapeutics
Procoagulant contaminants are a major contributor to hemostatic risk in human blood plasma-derived protein biotherapeutics. When plasma-derived biotherapeutics are contaminated with activated clotting factors like plasma kallikrein, FXIIa, or FXIa, this can result in a hemostatic insult with responses that are generally delayed. While these adverse events occur in less than 1% of individuals receiving IVIG therapy, any hemostatic risk must be mitigated for these drug products to be deemed both safe and effective.
Thorough analysis and removal of procoagulant contaminants is an essential part of plasma-derived biotherapeutics process development and maturation. A combined approach of high-resolution mass spectrometry (HRMS) and specific bioanalytical methods are required to verify removal of these contaminants from the product stream and to ensure product safety. Current procoagulant testing methods include:
- NaPTT clot-based assay – looks for generic intrinsic clotting activity
- PKA chromogenic assay – specific for FXIIa contamination
- FXIa chromogenic assay – specifically for FXIa contamination
- High Resolution Mass Spectroscopy (HRMS) – not generally viewed as being
quantitative - Thrombin generation assays (TGA) – specific for FXIa-like activity, sensitive to
FXIa levels not seen by HRMS
Challenge: Developing a validated and scalable process for a novel method of
IgG precipitation
Prolytix was presented with a novel plasma fractionation technology that utilized salt rather than ethanol to precipitate IgG. The technology was designed to not only take significantly less time than the Cohn process (48-72 hours vs. 7-10 days), but also to produce a significantly higher yield of IgG (>80% vs. 50-60%). Prolytix was charged with validating this fractionation process and developing a complete scalable process for IVIG production.
As Prolytix was developing the process for this client, we simultaneously tested for procoagulant contaminants, refining the process and removing contaminants in real time, shaving between three to six months from the client’s timeline. As this plasma fractionation methodology was newly patented, there was no existing literature to inform method development for removal of process contaminants.
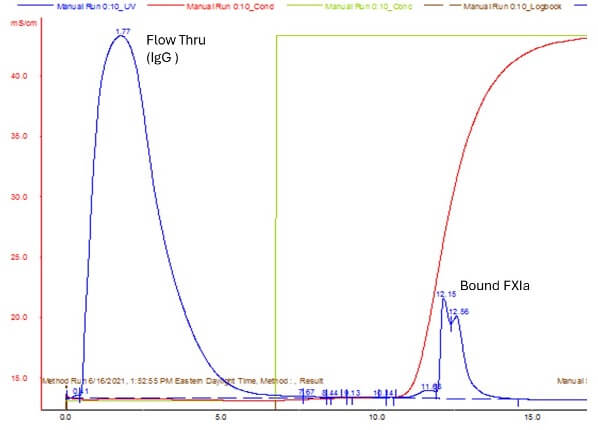
The first process we developed involved crude fractionation, depth/nominal filtration and IEX chromatography. The process utilized scalable equipment and was ready for pilot scale (50-100L) production. During release testing, we could not detect any clotting activity or FXIIa by PKA or NaPTT, but both HRMS and TGA assay indicated FXIa contamination. While the final formulation passed most European Pharmacopoeia (EP) metrics for safety and release, this FXIa contamination rendered the final product unacceptable for release.
Figure Two: FXIa contamination rendered the final formulation unfit for release
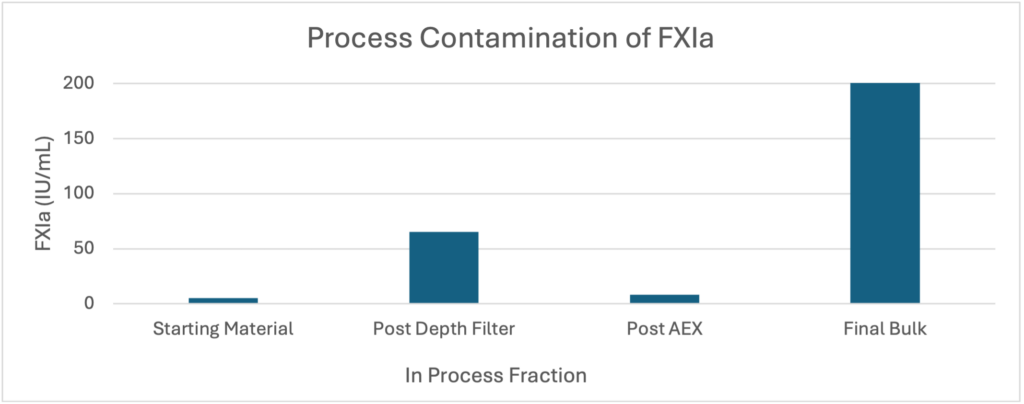
FXIa is a dimeric (80,000 Da subunits) coagulation enzyme that serves as an interface between the two primary coagulation pathways. FXIa is a robust enzyme with a long half-life in plasma. Small amounts of FXIa can cause serious thrombotic issues in an otherwise hemostatic patient. In this case, we hypothesized that the source of FXIa contamination was the depth filtration step, as we knew the filtration media, diatomaceous earth, to be an intrinsic activator.
Approach – Integrating a Polishing Step to Analyze and Remove Procoagulant
Contaminants
At first, we attempted to swap the filtration media in the depth filtration step for an alternative. The new batch, using a new depth filter, passed FXIa metrics, but failed NaPTT and PKA assays. Ultimately, swapping the depth filter material did appear to remove FXIa contaminants, but produced an even worse contaminant profile with plasma kallikrein activator.
Next, we considered the addition of an additional ion exchange (IEX) “polishing” step to remove FXIa during chromatography. The initial process contained a single anion exchange (AEX) chromatography step in flow through mode to positively select (>98%) contaminants. During method development, a cation exchange (CEX) step was included after the AEX step in a positive selection mode, however this step was removed as it markedly reduced he IgG yield.
It was determined that the best way to integrate a polishing step into the process would be to enable flow through of the product through the entire process. Since FXIa was only detectable in the final bulk product with highly sensitive methods, exogenous FXIa was spiked into the IEX flow through (containing 98+% pure IgG) to act as a standard for experimentation. The spiked material was subsequently passed over a weak cation exchanger (Figure 3A) and the IgG and FXIa were selectively eluted with a salt gradient which indicated a significant difference in affinity. This difference in affinity was exploited (Figure 3B) to increase the ionic strength of the initial anion exchange flow through to the point where IgG flowed through the cation exchanger while FXIa remained bound.
Figure 3: FXIa Removal with a cation exchanger
A. AEX flow through that has been dialyzed to reduce ionic strength, spiked
with exogenous FXIa and eluted with a 5 CV NaCl gradient
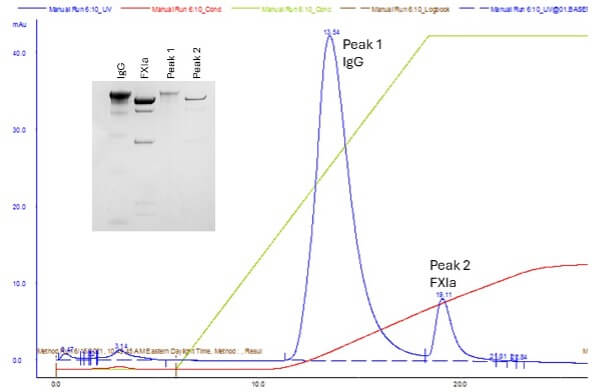
B. AEX flowthrough spiked with FXIa and loaded at 125 mM NaCl. FXIa is
subsequently eluted with a 0.5 M NaCl push. The FXIa is removed and the
product remains unbound in the flow through.
This proof-of-concept experiment with traditional ion exchange media allowed us to take advantage of a new technology in the form of an ion exchange filter. These filters are single use and are constructed from typical nominal filtration media (regenerated cellulose) that have been functionalized with an ion exchange moiety. These filters are ideal for the removal of trace contaminants and they allow for high flow rates with little to no loss of yield for the target molecule. When we adapted these filters into the process, this single change resulted in significantly higher yield (77%) and purity (99.7% pure IgG) than was achieved with a traditional resin-based polishing step. FXIa was effectively removed entirely from the process and the final material passed all EP metrics for safety. TGA and HRMS testing both indicated no detectable FXIa contamination.
Discussion
Procoagulant contaminants in a plasma-derived drug product pose a significant risk, and may not be detectable, even by powerful tools like HRMS. They can only be elucidated with sophisticated tests such as TGA, PKA, or NaPTT assays that effectively amplify their apparent activity. If procoagulant contamination is discovered in the process development of plasma-derived biotherapeutics, working with a CRO with significant analytical expertise, especially one that takes the time to understand the contaminant profile and where it is coming from, will pay off when formulating a strategy to remove it. Seemingly simple choices like the type of depth filtration media can have a dramatic effect on the procoagulant contaminant profile. Chromatography can be extremely useful, with many innovative solutions appearing on the market every day. Using an integrative approach to contaminant analysis (HRMS, TGA, etc.) is an excellent way to eliminate a procoagulant contaminant from a product stream.
The final yield of this process at bench scale was 75% IgG compared to the starting cryo-poor plasma. We also developed this process to be infinitely scalable with state-of- the-art technology, which we expect to produce yield gains of 5-10%. The new process consists of 8 steps and takes only 48 hours to complete compared to the 7-10-day cycle time for the Cohn process, resulting in significant cost savings and improved yields for IVIG manufacturers. Following Prolytix’s process development and validation work, this novel process for industrial production of an IVIG product that passes all EP standards for safety and purity is now being considered by a large scale IVIG manufacturer.
This project involved a significant research effort, so it was essential that the Prolytix team establish a strong trust-based relationship with the client. Throughout the 18 months of process development described in this case study, we had weekly meetings with the client, often providing daily updates to ensure that they had full transparency of both the progress we were making and the challenges we encountered. The client was a frequent visitor to our labs, and ultimately was extremely happy with the outcome of this work.
Prolytix has significant scientific expertise, and excels in the development and validation of novel processes and analytical methods. Our working style is highly collaborative — both internally and with the client — ensuring that we stay nimble and adjust quickly to accommodate client needs, deploying proprietary project management software to ensure that projects stay on track. At the same time, we have developed a reputation for being straightforward with clients about the scientific feasibility of projects, all of which has contributed to our 100% on time delivery average in 2023.
Interested in working with us? Get in touch with our experts!
